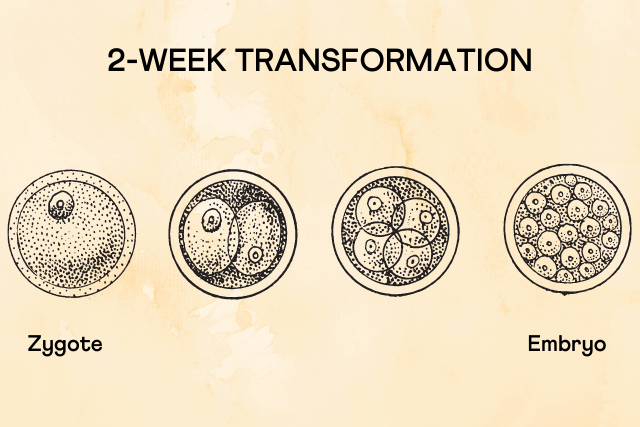Abstract
When we are born, we inherit (receive) the genes from our parents, but we don’t inherit their age.
In other words, how do older parents give rise to young offspring?
Scientists have tried to explain this by a process called “natural rejuvenation event.” This event sets our biological age to zero, which is the beginning of aging in mammals.
This event happens as the zygote develops to form an embryo. A zygote is a single-celled organism resulting from a fertilized egg. The zygote goes through a cellular multiplication process to give rise to the multi-celled embryo.
Researchers are studying the possible application of this “age reversal event” as a therapeutic intervention for age-related diseases, such as arthritis or Parkinson’s.
What is Aging?
Aging is something we all experience but actually know very little about. Hair greying, wrinkles formation, joint changes, etc., are signs of aging. Aging, on the other hand, encompasses all the processes that occur in the body that result in these signs – it is a combination of physiological changes in our bodies and environmental factors.
Aging eventually leads to physical denaturation, dysfunction, and ultimately mortality (death).
As we age, every cell in our body gets old due to the progressive accumulation of damage through the intervening years. But this cellular damage does not get passed on to human offspring to the next generation – the absence of this cellular damage in human offspring has baffled scientists for generations.
That’s because germline cells (sex cells – egg and sperm that form the embryo/zygote) rejuvenate in the offspring after conception.
Rejuvenation Event In The Embryo
During the initial stages of embryogenesis (the formation of the embryo), life hits a reset button — the rejuvenation event in the embryo. When an embryo is attached to the uterus, the rejuvenation event sets the developing embryo to grow at its youngest biological age – zero. This rejuvenation of the embryo marks the beginning of the mammalian age.
The epigenetic aging clock is a biochemical test used to measure age. It was developed with conserved cytosines – units in DNA whose methylation levels change with age. Experts use these epigenetic clocks to predict the approximate ages of mouse embryos at their early stages of development.
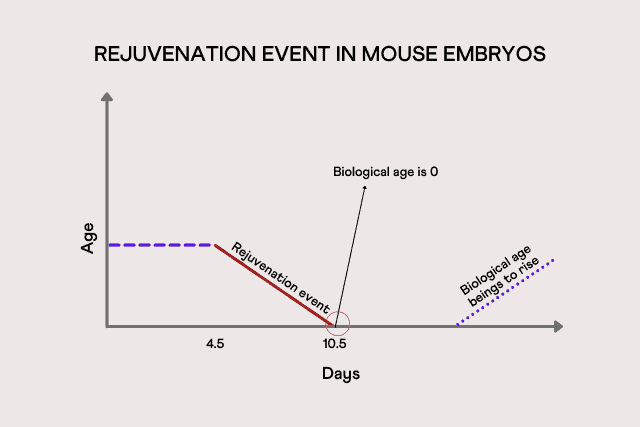
They found that the age of mouse embryos stayed constant for the first few days after fertilization. However, by around 6.5 to 7.5 days into development, the biological age of the embryos witnessed a dip, indicating that they are undergoing some type of rejuvenation event.
The researchers say that the biological age of mice is set to zero, somewhere between 4.5 to 10.5 days after fertilization.
Eventually, during development, the biological age begins to slowly rise. However, the point exactly at which this growth happens is still unclear.
At present, similar data for humans is unavailable as studying human embryos at such early stages is prohibited. Recent studies have shown that the epigenetic clocks in cord blood can be used to estimate gestational age at birth.
Potential Applications
When embryos can reset the biological clock, there is a hope to reverse the errors that occur due to cell damage, restoring the cells back to their agile state, eliminating signs of aging.
This hypothesis could possibly help researchers develop treatments for age-related diseases, such as arthritis or Parkinson’s – A paradigm shift in treating age-related illnesses.
Video
Summary
- The mystery behind how aged parents produce youthful offspring has puzzled researchers for a long time now.
- A recent study revealed a natural rejuvenation event during early embryogenesis that sets our biological age zero.
- The study noted that mouse embryos experience the rejuvenation event around 4.5 to 10.5 days after fertilization.
- Epigenetic aging clocks can help predict the embryos’ biological age and the onset of aging in mammals.
- This approach may pave the way for researchers to find treatment for age-related diseases by reversing the errors due to cell damage (using the same concept of age reversal seen in embryos).